What is the Lewis structure of lithium?
| lithium | 1 s 2 2 s 1 | 1 valence electron |
| beryllium | 1 s 2 2 s 2 | 2 valence electrons |
| nitrogen | 1 s 2 2 s 2 2 p 3 | 5 valence electrons |
| neon | 1 s 2 2 s 2 2 p 6 | 8 valence electrons |
What is the Lewis dot diagram for lithium?
Sep 02, 2013 · A step-by-step explanation of how to draw the Lewis dot structure for Li (Lithium). I show you where Lithium is on the periodic table and how to determine ho...
How to write Lewis dot structure?
3 rows · Mar 19, 2022 · March 19, 2022 by Saarah. Note: Lithium is in Group 1. Since it is in Group 1 it will have 1 ...
What are Lewis structures?
May 03, 2020 · The dots in the Lewis dot structure symbolize an atom's elements. Since Na^+ is a positive ion ...
What is the Lewis dot for lithium?
Jun 13, 2020 · Category: science chemistry. 4.4/5 (4,079 Views . 42 Votes) Note: Lithium is in Group 1. Since it ...
What is the Lewis dot structure of lithium?
0:191:17Lewis Dot Structure for Lithium (Li) - YouTubeYouTubeStart of suggested clipEnd of suggested clipAnd will put that one valence electron. Right next to the element symbol Li for lithium. So that'sMoreAnd will put that one valence electron. Right next to the element symbol Li for lithium. So that's the Lewis dot structure for lithium. This is dr. B and thanks for watching.
What is the Lewis dot structure for lithium fluoride?
0:211:42How to Draw the Lewis Dot Structure for LiF: Lithium fluoride - YouTubeYouTubeStart of suggested clipEnd of suggested clipSo the lithium transfers its one valence electron to the fluorine since the lithium. Lost anMoreSo the lithium transfers its one valence electron to the fluorine since the lithium. Lost an electron and electrons are negative the lithium's now has a positive charge.
How do you draw Lewis structures?
How to Draw a Lewis StructureStep 1: Find the Total Number of Valence Electrons. ... Step 2: Find the Number of Electrons Needed to Make the Atoms "Happy" ... Step 3: Determine the Number of Bonds in the Molecule. ... Step 4: Choose a Central Atom. ... Step 5: Draw a Skeletal Structure. ... Step 6: Place Electrons Around Outside Atoms.More items...•Jan 29, 2020
What is the correct Lewis structure for lithium sulfide?
0:001:30How to Draw the Lewis Dot Structure for Li2S : Lithium sulfide - YouTubeYouTubeStart of suggested clipEnd of suggested clipSo we'll put sulfur in the middle. And then lithium on either side. The theum is in Group 1 on theMoreSo we'll put sulfur in the middle. And then lithium on either side. The theum is in Group 1 on the periodic table that means it has one valence electron. And then sulfur that's in group 16.
What do Lewis structures show?
A Lewis Structure is a very simplified representation of the valence shell electrons in a molecule. It is used to show how the electrons are arranged around individual atoms in a molecule. Electrons are shown as "dots" or for bonding electrons as a line between the two atoms.Aug 15, 2020
What is a single Lewis structure?
In the Lewis model, a single shared pair of electrons is a single bond. Each Cl atom interacts with eight valence electrons total: the six in the lone pairs and the two in the single bond.Jun 5, 2019
How do you determine the best Lewis structure?
Lewis structures are preferable when adjacent formal charges are zero or of the opposite sign. When we must choose among several Lewis structures with similar distributions of formal charges, the structure with the negative formal charges on the more electronegative atoms is preferable.
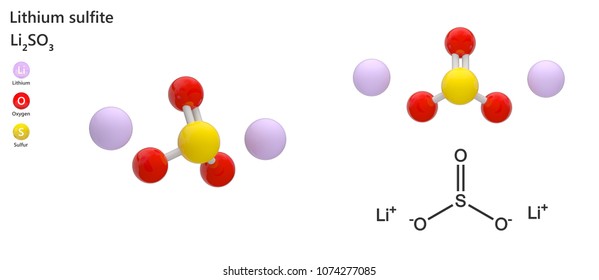
What is the formula for lithium sulfate?
Li2SO4Lithium sulfate / Formula
What is the formula of lithium chloride?
LiClLithium chloride / Formula
What is the formula for lithium sulfite?
Li2SO3Lithium sulfite / Formula
How many valence electrons does lithium have?
Note: Lithium is in Group 1. Since it is in Group 1 it will have 1 valence electrons. When you draw the Lewis structure for Lithium you'll put one "dot" or valance electron next to the element symbol (Li).
What is the Lewis symbol for carbon?
Lewis Symbols For example, the Lewis symbol of carbon depicts a “C' surrounded by 4 valence electrons because carbon has an electron configuration of 1s22s22p2. The Lewis symbol for carbon: Each of the four valence electrons is represented as a dot. Is Lithium a cation or anion?
What is lithium cation?
Lithium Cation is a monovalent cation that is metabolized much like sodium and is important in many cellular functions inside or on the surface of cells. Lithium was used during the 19th century to treat gout. Lithium salts such as lithium carbonate (Li2CO3), lithium citrate, and lithium orotate are mood stabilizers.
Who invented the electrolyte for lithium / thionyl chloride?
Jean-Paul Gabano, "Electrolyte for a lithium / thionyl chloride electric cell, a method of preparing said electrolyte and an electric cell which includes said electrolyte." U.S. Patent US4375502, issued 0000.
What is lithium salt used for?
Lithium salts such as lithium carbonate (Li2CO3), lithium citrate, and lithium orotate are mood stabilizers. They are used in the treatment of bipolar disorder, since unlike most other mood altering drugs, they counteract both mania and depression. Lithium can also be used to augment other antidepressant drugs.
What is GSK-3B?
GSK-3 is known for phosphorylating and thus inactivating glycogen synthase. GSK-3B has also been implicated in the control of cellular response to damaged DNA. GSK-3 normally phosphorylates beta catenin, which leads to beta catenin degratation.
Does lithium change the currents of glutamate receptors?
Lithium has been shown to change the inward and outward currents of glutamate receptors (especially GluR3), without a shift in reversal potential.
Is lithium good for bipolar?
Although lithium has been used for over 50 years in treatment of bipolar disorder, the mechanism of action is still unknown. Lithium 's therapeutic action may be due to a number of effects, ranging from inhibition of enzymes such as glycogen synthase kinase 3, inositol phosphatases, or modulation of glutamate receptors.
Is HMDB a free resource?
LICENSE. HMDB is offered to the public as a freely available resource . Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page).